- Visibility 14 Views
- Downloads 2 Downloads
- DOI 10.18231/j.ijooo.2021.009
-
CrossMark
- Citation
Management of OSSN by different modalities of treatment
- Author Details:
-
Arif Khan
-
Anju Kochar *
-
Nabab Ali Khan
Introduction
The term Ocular Surface Squamous Neoplasia (OSSN) was coined by Lee and Hirst[1] in 1995. OSSN is an encompassing term for precancerous and cancerous epithelial lesions of the conjunctiva and cornea. It includes the spectrum of dysplasia, CIN, and invasive SCC.[2] Previously used terms for this condition include intraepithelial epithelioma, Bowens disease, and Bowenoid epithelioma.[3] It is the most common non-pigmented malignancy of the ocular surface.[4]
The incidence of OSSN is about 0.02 to 3.5 cases per 1,00,000 population per year and shows ethnic and geographic variations.[5] With every 100 decline in latitude, there is an increase in the incidence of ocular Squamous Cell Carcinoma by 49%.[6] The average age of presentation is usually the sixth and seventh decades of life. However, in immunocompromised individuals, OSSN may occur at a younger age.[7]
The precise etio-pathogenesis of these lesions is not very clear but various postulated risk factors include fair skin, male sex and advancing age,[8] exposure to ultraviolet radiations, cigarette smoking,[9] organ transplant, autoimmune conditions,[10] infection with Human Papilloma virus (HPV)[11] and Human Immunodeficiency Virus (HIV). Several studies have reported a dramatic increase in the incidence of OSSN following the outbreak of HIV.[12] Systemic associations of the development of OSSN include xeroderma pigmentosum and Papillon–Leferve syndrome.[13]
OSSN are important because they mimic many common indolent lesions like pterygium and have a potential for causing ocular and systemic morbidity and mortality.[14] OSSN usually presents either as a fleshy, gelatinous, elevated lesion or as a sessile, papillomatous lesion. It usually starts in interpalpebral conjunctiva and then grows and straddles the limbus and then may or may not involve the cornea. Most often vision is not affected unless the lesion is encroaching onto the pupillary area. OSSN patients usually present with a swelling, redness, irritation and slightly elevated and have a pearly grey appearance with tufts of vessels commonly known as sentinel vessels, with or without well-defined borders.[13] OSSN Metastasis occurs in 2 mm thickness or those located in fornix, caruncle, or orbit are at highest risk for metastasis.[10] Advanced cases can infiltrate the cornea and sclera[15] and rarely the tumour may extend into the orbit causing proptosis.
Corneal OSSN lesions are pre invasive, with a mottled ground glass sheet appearance which is opalescent. They have sharply defined fimbriated borders, the convex leading edge spreads in an arc away from the limbus and often white dots are present over the grey epithelium. They are usually avascular. These lesions are typically indolent, slow growing and prone to recurrence.
The various diagnostic test available for these neoplasm are Rose Bengal staining, Exfoliative and Impression Cytology, Histopathology, Anterior Segment Optical Coherence Tomography (AS-OCT), Ultrasonic biomicroscopy and Confocal Microscopy. AS-OCT and confocal microscopy, hold promise in invivo differentiation of intraepithelial neoplasia from invasive squamous cell carcinoma.[16]
OSSN has a relatively high recurrence rate after treatment and may metastasize. These tumours are considered to be low grade malignancies but invasive lesions can spread to the globe or orbit.[17]
The standard modality for treatment of OSSN has been wide surgical excision with “no-touch” technique[18] and adjunctive cryotherapy. However, due to high recurrence rates ranging from 5% to 66% after surgical excision,[19] nonsurgical management with topical chemotherapeutic agents (5-fluorouracil) and mitomycin C (MMC) has become the preferred choice for management of OSSN.[20] Recently Interferon-a 2b (IFNα2b) as well as pegylated IFNs are regularly being used in a select subsets of OSSN for treatment.[14]
Materials and Methods
This study is a histopathological, prospective biopsy specimen series carried out in the Department of Ophthalmology, S.P. Medical College & Associated Group of Hospitals, Bikaner in collaboration with Pathology Department.
Method
After permission from institutional review board, the study was conducted as histopathological hospital based prospective comparative interventional case series carried out in the Department of Ophthalmology, S.P. Medical College and associated group of hospital from December 2017 to December 2019.
The study included 22 consecutive cases of OSSN of either age and sex.
A detailed slit lamp examination of all eyes was done. Patients’ demographic, pathologic and clinical data before treatment were recorded, these data included age, sex, occupation, history, location, shape, size, border, margin, ulceration, induration and mobility over underlying surface of tumour and visual acuity. Pre operative investigation like HB, BT, CT, viral marker, urine sugar & albumin were done before surgery. All patient of OSSN were screened for HIV. According to nature and size of conjunctival growth/neoplasia two categories were formed.
If the growth was small or medium size (covering less than half of cornea) and appeared to be malignant clinically then surgically excision was done.
In case of large growth (covering more than half of the cornea) incisional biopsy was taken.
There was only one patient in which surgical excision was done, in which growth was more than 4 clock hours because she did not tolerate topical chemotherapy. In all others cases surgical excision of growth with wide margin was done for small tumours.
Cryotherapy by double freeze thaw technique was applied in cases following excision.
Excisional Biopsy specimen was sent to the Department of Pathology, for histopathological examination. After confirmation of OSSN on histopathological examination, growths with normal margins on histopathology were not treated further, while those with malignant cells along the margins of excised tissue were given adjuvant mitomycin drops post operatively four cycle 0.04% (0.4mg/ml) Mitomycin eye drop was given, four times a day for four continuous days per week followed by alternate one week off and one week on (total duration of treatment was 8 weeks).
In our study three groups are formed based on different modalities for treatment of OSSN-
Group -1: Surgery plus MMC (0.04%) 4 cycles
Group -2: Surgery
Group -3: Only MMC (0.04%) 4 cycles
Method of MMC Eye drop formation
10 mg MMC was mixed with 10 ml. distilled water to form 1mg/ml solution. 2 ml of this solution was than mixed with 3ml distil water to form 0.4mg/ml (0.04%) solution. This solution was used with 0.5% carboxy methyl cellulose to decrease MMC related side effects.
Post operative treatment was moxifloxacin + dexamethasone eye drops, moxifloxacin eye ointment, 0.5% carboxy methyl cellulose for soothing effect, Tab. Vitamin C 500mg used for early wound healing.
Follow up was done at post operative 1st day, 1st week, 3rd week, 3 months there after 6 monthly intervals and earlier if the patient needed.
Data collection & analysis
Data thus collected were entered into Microsoft Excel sheet. Statistical analyses were performed using the SPSS ver. 15.0 (SPSS Inc., Chicago, IL, USA). Results were analyzed as tables, proportions in case of qualitative data, mean and standard deviation in case of quantitative data. p-value of less than 0.05 was considered to be statistically significant.
Results
A total of 22 patients completed the study. Demographic data of the patients and clinical features were following
Mean age ± SD: 54.10±13.17
Growth of mass was the chief complaint in all 22 patients (100%).Associated complaints like foreign body sensation were seen in 17 patients(77%), watering in 14 patients(64%), followed by brownish discoloration of eye was found in 5 patients (23%). None of patient complained with diminuation of vision and pain. Mean duration of symptoms was 10 months. In all patients tumour located within palpebral aperture at limbus with temporal/nasal ratio 2:1.
The ratio of male/female was 16/6 (2.7/1) and rural/urban: 17/5 (3.4/1).
Maximum number of patients (12) in age groups 41-60 years 8 case in 61-80 years age groups and 2 case was below 40 years. Farmers were most commonly affected by occupation with total 14 cases while second most commonly affected were house wife. Student, truck driver and servicemen have single case.
Maximum number of patients were farmers or those engaged in outdoor occupation.
The gross appearance of ocular surface squamous neoplasia in our study population was also studied in terms of Surface, Margins, Mobility, Laterality, corneal involvement, feeding vessels and whether it was excised or not. 45.45% neoplasia had smooth surface on examination, 59.09% had regular margins, all were immobile, 59.09% of the patients had right eye involvement, 95.46% presented with corneal involvement, 86.36% showed presence of feeding vessels and 95.46% patients with ocular surface squamous neoplasia showed positive rose Bengal staining. Nodular elevation (50%) was most commonly found shape followed diffuse fungating (27.27%) growth. Details of gross morphological appearance described in [Table 1]. Irregular surface had uneven topography while rough surface indicates that it was grainy in apprearance.
Out of 22 cases 21 patients were surgically excised and post operative 0.04% mitomycin (group 1) were given in 15 patients which had no recurrence while remaining 6 case were kept on observation after surgically excision (group 2) whose histopathology reports showed tumour free margins. However in spite of these reports one of those 6 patients had recurrence at same location which was treated according to group 1 plan. Remaining one case present with diffuse corneal involvement and growth present at two site which was already dignosed case of xeroderma pigmentosa and loss of other eye due to squmous cell carcinoma. In this patient incisional biopsy was taken and after histopatholoical confirmation 0.04% mitomycin-c was given and completely cured with no recurrence (group 3). Details of various treatment modilities are shown in [Table 2].
On Histopathological examination 15 cases 68.18% were reported with squamous cell carcinoma, 5 cases (22.73%) with severe dysplasia and two cases (9.09) with carcinoma in-situ. One OSSN patient had dual findings of Squmous. cell carcinoma + malignant melanoma of conjunctiva. One patient of OSSN was a confirmed case of AIDS.
|
Surface |
Frequency (N=22) |
Percent |
|
Smooth |
10 |
45.45 % |
|
irregular |
8 |
36.36 % |
|
Rough |
4 |
18.19% |
|
Margin |
||
|
regular |
13 |
59.09 % |
|
Irregular |
9 |
40.91 % |
|
Mobility |
||
|
Absent |
22 |
100.00 % |
|
Present |
00 |
00.00 % |
|
Laterality |
||
|
Right eye |
13 |
59.09 % |
|
Left eye |
9 |
40.91 % |
|
Corneal involvement |
||
|
Absent |
01 |
4.54 % |
|
Present |
21 |
95.46 % |
|
Feeding vessels |
||
|
Absent |
03 |
13.63 % |
|
Present |
19 |
86.36 % |
|
Excision |
||
|
No |
01 |
4.54 % |
|
Yes |
21 |
95.46 % |
|
Rose bengal staining |
||
|
Negative |
01 |
4.54 % |
|
Positive |
21 |
95.46 % |
|
Modality used |
Cured |
Recurrence |
|
Surgery+ MMC (0.04%) 4 CYCLE (N=14) |
14(100.00%) |
0(0.00%) |
|
Surgery (N=7) |
6(85.71%) |
1(14.28%) |
|
MMC (0.04%) 4 cycle (N=1) |
1(100.00%) |
0(0.00%) |
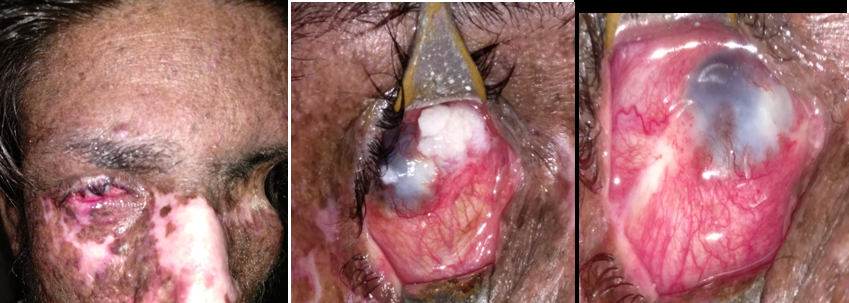
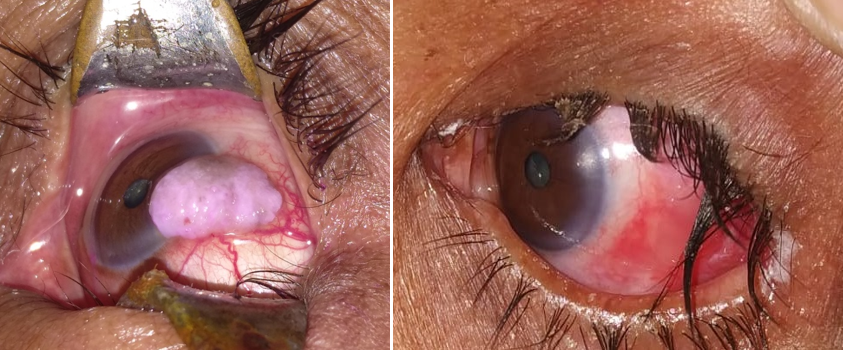
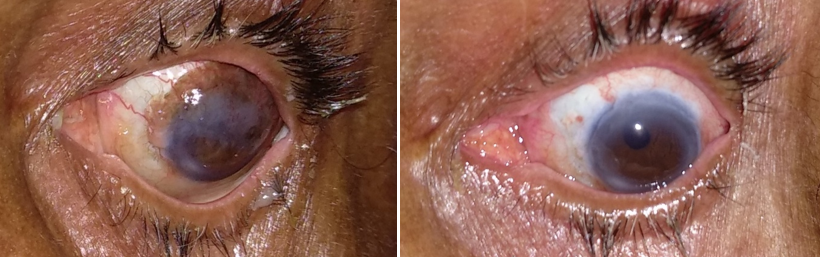
Discussion
Malignant tumours were more prevalent in males because males are usually engaged in outdoor activities resulting in increased exposure to risk factor like UV rays and other outdoor occupational hazards.
75% of our study population were resident of rural areas. Frequency of these lesions in rural areas, can be attributed to outdoor occupations. Farmers had the highest prevalence of OSSN as they are more prone to exposure of risk factors (sun light UV rays). The occurrence of ocular surface neoplasia according to occupation was observed to be statistically significant (p<<0.05). The incidence of OSSN is associated with solar UV exposure. Newton et al.[6] showed that the incidence of OSSN declined by 49% for each 10° increase in latitude, from more than 12 cases/million/year in Uganda [latitude 0.3°] to less than 0.2 million/year in the United Kingdom [latitude >50°]. A case–control study[21] of OSSN in Uganda confirmed that the risk of OSSN was higher in those exposed to sunlight and it increased in a dose-dependent fashion. In Uganda, they found an increased incidence of p53 mutations, known to be associated with sun exposure.[22]
One OSSN patient had Xeroderma pigmentosa while another one was on anti retroviral treatment, both of which are considered as risk factor of OSSN.
The cause of OSSN is thought to be. OSSN has been increasing in incidence since the HIV pandemic, and this strongly suggests that HIV increases the risk for OSSN.[23] A case–control study[24] showed that conjunctival malignancy was the first presenting sign for AIDS in 50% of their patients. A retrospective study by Karp et al. [25] found that for patients less than 50 years of age diagnosed with OSSN, 50% were HIV positive. Therefore, when younger patients present with OSSN, we suggest that they should be screened for HIV. OSSN can occur at any CD4+ T cell count; however, in the few studies [26], [27] in developing, it has been reported that the OSSN can occur CD4+ T cell count less than 200 cell/microlitre µl.
Furthermore, patients with xeroderma pigmentosa are incapable of repairing damage from the sun, and they present with more aggressive cases of OSSN and at younger ages.[28] It is thought that solar exposure aids in the development of OSSN through DNA damage and with the interaction of other factors leading to the increased risk of tumour genesis.[29]
With respect to morphology nodular elevation (50%) was found in majority of OSSN patients in our study which is a finding similar to study done by Rachna Meel et al [30] (2017) who found nodular elevation in the majority of cases.
45.45% OSSN had smooth surface with irregular margin and right eye involvement in 59% (no relevant significance), all were immobile and almost all had corneal involvement and showed positive rose bengal staining. These are typical finding of OSSN.
At our center, we have devised our own clinical classification scheme for OSSN (small, large and diffuse) based on the clock hours of limbal involvement and maximal basal diameter, which is used to decide treatment for tumors. In our study we used three modalities for treatment of OSSN- surgery, surgery plus MMC (0.04%) 4 cycles and only MMC (0.04%) 4 cycles whereas Ruchi Mittal (2013)[16] used Surgery, chemotherapy and immunotherapy as treatment modalities. In J.M. Manoher et al[31] (2016) study MMC was used as sole therapy for diffuse OSSN and with surgical excision in localized OSSN. Choice of treatment depends upon tumor extent as well as surgeon’s decision.
MMC is a potent cytotoxic agent isolated from Streptomyces caespitosus. It undergoes metabolic activation to become an alkylating agent that is cytotoxic to both proliferating and non-proliferating cells. It functions in all phases of cell cycle, especially in rapidly dividing cells. It acts by inhibiting DNA synthesis preferably in G1 and S phase. One of the major limitations of topical MMC therapy is the lack of a recommended optimal dose and duration of treatment. The lack of major complications is supportive of this treatment regime. Limbal toxicity from MMC has been reported, hence, a one week-on one week off regime is used to minimise toxicity to normal healthy ocular surface and periocular tissues, especially limbal stem cells. MMC was available free of cost at our centre hence it was used, while Interferon alpha 2b was not used due to logistic problems (higher cost and availability issues).
Out of 14 patients 1 patient was discontinued from MMC after only 2 cycles due to side effect related to MMC. Whereas J.M. Manoher et al (2016) found ocular irritation and mild conjunctival hyperemia were observed in 4 eyes during treatment with MMC. Excision remains an important step in management of localised carcinoma in situ. Excision allows an immediate histopathological diagnosis and excludes life-threatening invasive malignancies such as SCC or amelanotic malignant melanoma. It also helps to diagnose masquerading lesions such as viral papilloma, where MMC is not effective, and lesions keratoacanthoma and solar keratosis, where MMC is not necessary. Surgical debulking of the lesion makes adjuvant treatment more effective, as MMC is being utilized against a lower tumour load.
When treating disease involving more than six clock hours of limbus, excision is potentially hazardous due to the risk of limbal stem cell failure and so is avoided in our study and planned for incisional biopsy. MMC is used as sole therapy for cases with diffuse limbal involvement for more than six clock hours.
Indications for topical chemotherapy in our study were:-
Diffuse limbal involvement for more than six clock hours.
Residual tumour after excision.
Patient not fit for surgery.
Protocol for topical MMC (rule of 4) in our study
0.04% (0.4 mg/ml)
4 times a day
4 continuous days in one week
4 cycle were given
Alternate one week off and one week on treatment (total time 8 weeks)
Conclusion
To conclude, we present the clinico-demographic profile of OSSN that these tumour are more prevalent in older age groups with male predominance, residing in rural areas and farmers by occupation. Surgery plus MMC were the most effective mode of treatment in these neoplasia. These types of tumours can be treated with only mitomycin-c in cases where surgery was challenging. Young patients presenting with OSSN must be evaluated for HIV since HIV increase the risk of OSSN.
Abbreviations
OSSN: Ocular Squamous surface Neoplasia, SCC: Squamous cell Carcinoma, HPV: Human Papilloma virus, HIV: Human Immunodeficiency Virus, AS-OCT: Anteror Segment OCT, INF α2b: Interferon Alpha- 2b, CIN: Conjunctival Intraepithelial Neoplasia, MMC: Mitomycin: C, UBM: Ultrasound bio Microscopy, HB: Hemoglobin, BT: Bleeding Time, CT: Clotting Time.
Conflicts of Interest
All contributing authors declare no conflicts of interest.
Source of Funding
None.
References
- GA Lee, LW Hirst. Ocular surface squamous neoplasia. Surv Ophthalmol 1995. [Google Scholar] [Crossref]
- J Pe’er. Ocular surface squamous neoplasia. Ophthalmol Clin North Am 2005. [Google Scholar]
- L D Pizarello, F A Jakobeic, FA Jakobeic. Bowens disease of the conjunctiva: A misnomer. Ocular and Adnexal Tumors 1978. [Google Scholar]
- MV Cicinelli, A Marchese, F Bandello, G Modorati. Clinical Management of Ocular Surface Squamous Neoplasia: A Review of the Current Evidence. Ophthalmol Ther 2018. [Google Scholar] [Crossref]
- J Yang, CS Foster. Squamous Cell Carcinoma of the Conjunctiva. Int Ophthalmol Clin 1997. [Google Scholar] [Crossref]
- R. Newton, G. Reeves, V. Beral, J. Ferlay, DM Parkin. Effect of ambient solar ultraviolet radiation on incidence of squamous-cell carcinoma of the eye. Lancet 1996. [Google Scholar] [Crossref]
- Y Porges, G M Groisman. Prevalence of HIV With Conjunctival Squamous Cell Neoplasia in an African Provincial Hospital. Cornea 2003. [Google Scholar] [Crossref]
- S Gichuhi, E Macharia, J Kabiru, A M’bongo Zindamoyen, H Rono, E Ollando. Clinical Presentation of Ocular Surface Squamous Neoplasia in Kenya. JAMA Ophthalmol 2015. [Google Scholar] [Crossref]
- C Napora, E J Cohen, G I Genvert, A C Presson, J J Arentsen, R C Eagle. Factors associated with conjunctival intraepithelial neoplasia: A case control study. Ophthalmic Surg 1990. [Google Scholar]
- C L Shields, J L Chien, T Surakiatchanukul. Conjunctival tumors: review of clinical features, risks, biomarkers, and outcomes—the 2017 J. Donald M. Gass Lecture. Asia Pac J Ophthalmol (Phila) 2017. [Google Scholar]
- S Gichuhi, S Ohnuma, MS Sagoo, MJ Burton. Pathophysiology of ocular surface squamous neoplasia. Exp Eye Res 2014. [Google Scholar] [Crossref]
- M Guech-Ongey, EA Engels, JJ Goedert, RJ Biggar, SM Mbulaiteye. Elevated risk for squamous cell carcinoma of the conjunctiva among adults with AIDS in the United States. Int J Cancer 2008. [Google Scholar] [Crossref]
- SG Honavar, FP Manjandavida. Tumors of the ocular surface: A review. Indian J Ophthalmol 2015. [Google Scholar] [Crossref]
- S Gupta, R Sinha, N Sharma. Titiyal JSOcular Surface Squamous Neoplasia. DJO 2012. [Google Scholar]
- D. H. Nicholson, J. Herschler. Intraocular Extension of Squamous Cell Carcinoma of the Conjunctiva. Arch Ophthalmol 1977. [Google Scholar] [Crossref]
- R Mittal, S Rath, G K Vemuganti. Ocular surface squamous neoplasia – Review of etio-pathogenesis and an update on clinico-pathological diagnosis. Saudi J Ophthalmol 2013. [Google Scholar] [Crossref]
- . Tananuvat N. and Lertprasertsuke N. Ocular Surface Squamous Neoplasia, Intraepithelial Neoplasia, Dr. Supriya Srivastava (Ed.), In-tech. Rijeka, Croatia. http://www.intechopen.com/books/intraepithelialneoplasia/ocular-surface-squamous-neoplasia. . [Google Scholar]
- J A Shields, C L Shields, P De Potter. Surgical management of conjunctival tumors. The 1994 Lynn B. McMahan Lecture. Arch Ophthalmol 1997. [Google Scholar]
- G Tabin, S Levin, G Snibson, M Loughnan, H Taylor. Late Recurrences and the Necessity for Long-term Follow-up in Corneal and Conjunctival Intraepithelial Neoplasia. Ophthalmol 1997. [Google Scholar] [Crossref]
- AA Nanji, CS Moon, A Galor, J Sein, P Oellers, CL Karp. Surgical versus Medical Treatment of Ocular Surface Squamous Neoplasia. Ophthalmol 2014. [Google Scholar] [Crossref]
- K. M. Waddell, S. Lewallen, S. B. Lucas, C. Atenyi-Agaba, C. S. Herrington, G. Liomba. Carcinoma of the conjunctiva and HIV infection in Uganda and Malawi.. Br J Ophthalmol 1996. [Google Scholar] [Crossref]
- C Ateenyi-Agaba. TP53 mutations in squamous-cell carcinomas of the conjunctiva: evidence for UV-induced mutagenesis. Mutagenesis 2004. [Google Scholar] [Crossref]
- N A V Beare, N H Batumba. The impact of HIV on sub-Saharan African eye departments. Eye 2006. [Google Scholar] [Crossref]
- Y Porges, GM Groisman. Prevalence of HIV With Conjunctival Squamous Cell Neoplasia in an African Provincial Hospital. Cornea 2003. [Google Scholar] [Crossref]
- C L Karp, I U Scott, T S Chang, S C Pflugfelder. Conjunctival intraepithelial neoplasia. A possible marker for human immunodeficiency virus infection?. Arch Ophthalmol 1996. [Google Scholar]
- K M Waddell, R G Downing, S B Lucas, R Newton. Corneo-conjunctival carcinoma in Uganda. Eye 2006. [Google Scholar] [Crossref]
- TG Pradeep, SB Gangasagara, GB Subbaramaiah, MB Suresh, N Gangashettappa, R Durgappa. Prevalence of Undiagnosed HIV Infection in Patients With Ocular Surface Squamous Neoplasia in a Tertiary Center in Karnataka, South India. Cornea 2012. [Google Scholar] [Crossref]
- N Gupta, R Sachdev, R Tandon. Ocular surface squamous neoplasia in xeroderma pigmentosum: clinical spectrum and outcome. Graefes Arch Clin Exp Ophthalmol 2011. [Google Scholar] [Crossref]
- G Cervantes, AA Rodríguez, A G Leal. Squamous cell carcinoma of the conjunctiva: clinicopathological features in 287 cases. Can J Ophthalmol 2002. [Google Scholar] [Crossref]
- R Meel, R Dhiman, M Vanathi, N Pushker, R Tandon, S Devi. Clinicodemographic profile and treatment outcome in patients of ocular surface squamous neoplasia. Indian J Ophthalmol 2017. [Google Scholar] [Crossref]
- J M Manoher, Praveena, A Kochar, A Chauhan. Topical 0.02% Mitomycin C for management of primary corneal-conjunctival intraepithelial neoplasia as primary therapy: A long term follow up. IAIM 2016. [Google Scholar]
